Rapid and cost-effective detection of specific sequences of DNA or RNA is an urgent need as COVID19 pandemics have demonstrated. Since more than 15 years we are pursuing this goal.
Electrochemical hybridization-based biosensors, also known as genosensors, are a low-cost, rapid and miniaturizable strategy to fulfil the desired criteria for a point-of need device (ASSURED-affordable, sensitive, specific, user-friendly, rapid and robust, equipment-free and delivered to those who need it). A detailed explanation on this strategy has been recently compiled (Understanding the factors affecting the analytical performance of sandwich-hybridization genosensors on gold electrodes
We have first described that p-aminothiophenol as a better diluent/blocking thiol than the widely used mercaptohexanol to construct genosensors on Au-based electrodes due to its aromatic and conducting nature (10.1021/acs.analchem.5b02271). Besides, monolayers of thioaromatic compounds with covalent linkage of capture DNA gives the best analytical performance when using SAM on Au to build the genosensor (10.1016/j.bios.2017.02.017)
A step further toward an integrated genosensor where amplification and detection is performed on the same device requires the use of isothermal DNA amplification strategies. Helicase dependent amplification (HDA) was successfully combined with several electrochemical genosensing showing similar detectability (30 copies) than PCR for the detection of Mycobacterium tuberculosis in clinical specimen (10.1016/j.bios.2014.12.029). We also used recombinase polymerase amplification (RPA) (10.1002/celc.201801208)
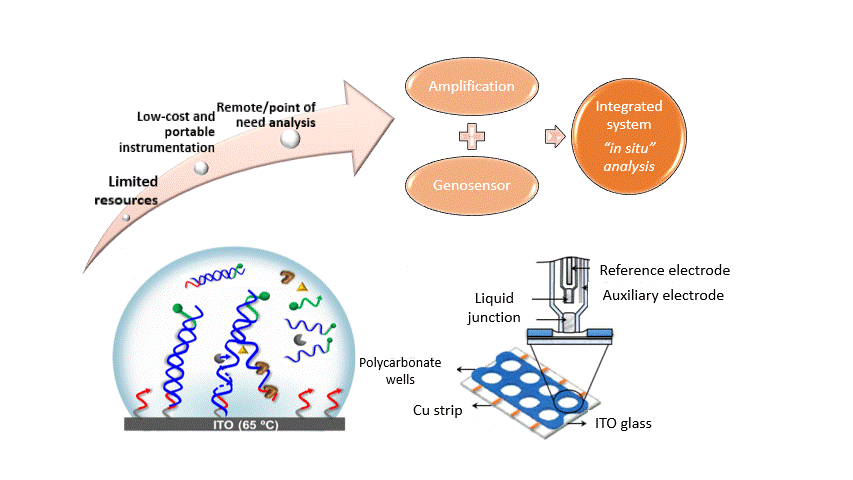
We have demonstrated the feasibility of performing the HDA on an ITO chip with in-situ electrochemical detection. Since the enzymatic product is colored both electrochemical and spectrophotometric measurements were compared obtained a ten-fold improved detection with the electrochemical detection (10.1039/C7CC05128J). Currently we are exploring other isothermal amplification strategies.
LIQUID BIOPSY: detection of LONG-NON CODING RNAS as cancer biomarkers
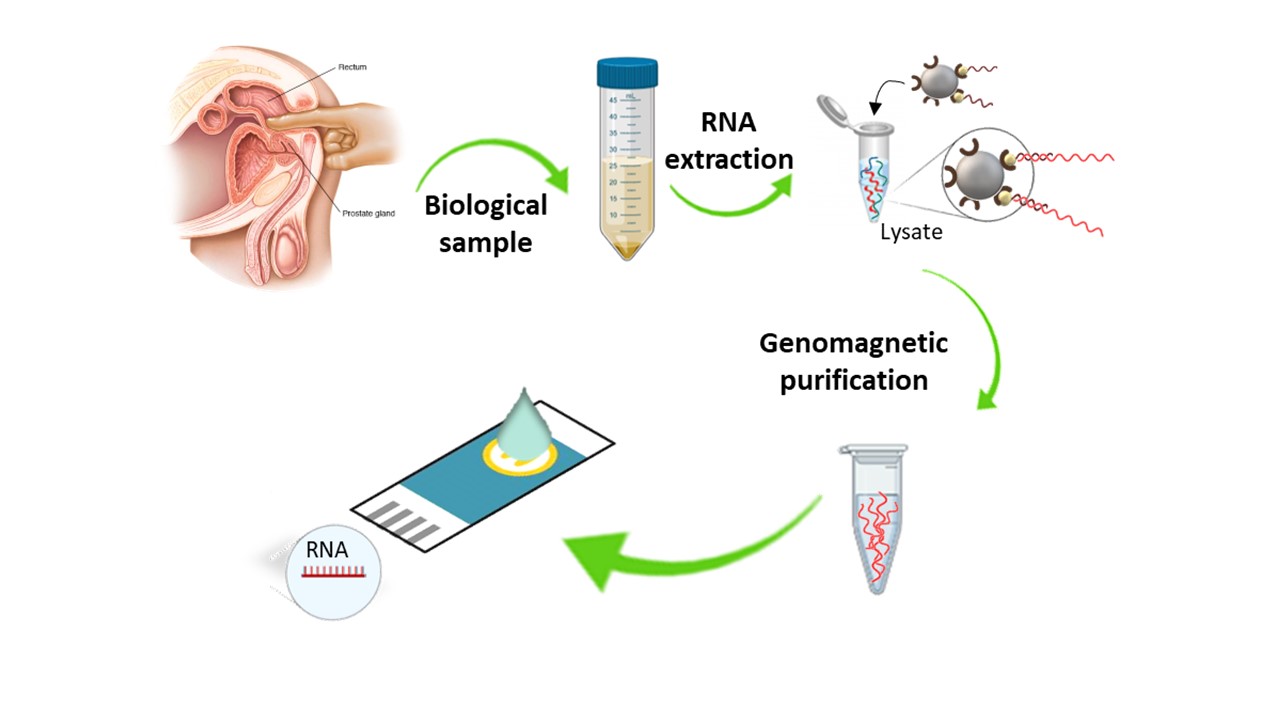
Cancer diagnosis ultimately relies on confirmatory biopsies, which are so invasive that cannot be repeated to monitor the progression/regression of the disease. This is the reason behind the concept of liquid biopsy, the development of analytical tools to detect biomolecules such as proteins, circulating DNA, circulating tumor cells, microRNA, lncRNA, exosomes, microvesicules in biological fluids that serve as disease biomarkers to eliminate invasive biopsies
Non-coding nucleic acids were regarded until the new century as «dark/trash matter» but the advances in massive sequencing and analytical methods of detection have revealed significant roles in the cellular metabolism. Both short (microRNA) and long non-coding RNAs are being associated to tumorigenesis because their level of expression is altered in cancer patients opening the way to become a tumor biomarker. Detection of both types of RNA has its own difficulties beyond those we have already addressed for detection of specific DNA sequences. They are frequently in extremely low levels (fM or lower).
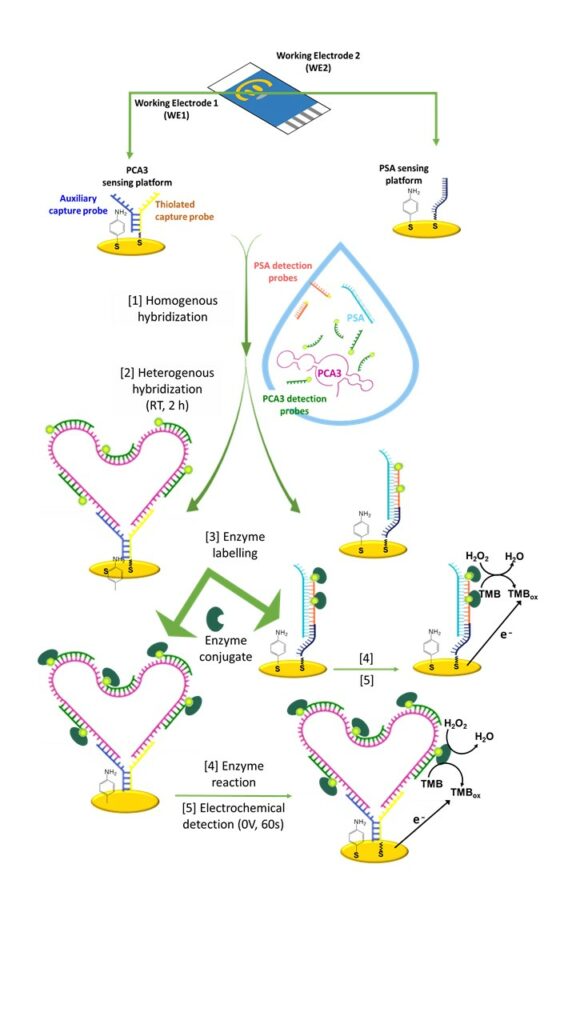
We have designed a dual genosensor for the specific detection of PCA3 lncRNA along with a reference mRNA for normalization in cell lines and urine from patients with prostate cancer.